 
Updates to the SmPC and PIL to include information about receiving a heterologous booster dose in individuals aged 18 years and over. The GB Conditional Marketing Authorisation has been converted to a full Marketing Authorisation. This update reflects the most up to date information about these known side effects. 
There has also been an update to the product information for the Comirnaty vaccines on myocarditis and pericarditis after a third dose and in the age group 5-11 years. Product information has been added for a new presentation of the vaccine for use in those aged 6 months to 4 years.Ĭomirnaty 10 micrograms concentrate for dispersion for injection (one of the licensed Comirnaty vaccines produced by Pfizer/BioNTech) has been approved for use as a booster for 5- to 11-year-olds. Updated the Comirnaty purple, orange and maroon cap SmPCs and PILs. & longer-term clinical trial follow-up data in 5-11 year olds. Updated SPCs and PILs for all Comirnaty products, to include: the uncommon side effect ‘dizziness’ Updated Links and hyperlinks in ARCHIVE: Information for UK recipients on Pfizer/BioNTech COVID-19 vaccine (Regulation 174) attachment Safety and immunogenicity information in the SmPC has been updated, based on six months post-booster dose follow-up data in recipients aged 16 year and older from studies C4591001 and C4591031. The MHRA regularly publishes reports of the safety of the COVID-19 vaccines. Please regularly check this information as it is often updated. The information for healthcare professionals and UK recipients on using the vaccine safely has been updated as new data have become available and this will continue under the MA. The GB CMA was converted to a full MA on 9 November 2022. The CMA issued by the EMA has continued to have effect in Northern Ireland since 21 December 2020 and was converted to a full Marketing Authorisation (MA) on 10 October 2022. 
The original conditional Marketing Authorisation (CMA) was issued by the European Medicines Agency (EMA) on 21 December 2020 and was automatically converted to a GB CMA on 1 January 2021. The archived product information for the COVID-19 Vaccine Pfizer/BioNTech authorised under Regulation 174 can also be found on this page. Supply of the vaccine is now in accordance with the Marketing Authorisation with all remaining Regulation 174 stocks expiring at the end of February 2022. Initially, the COVID-19 Vaccine Pfizer/BioNTech was supplied in the UK on a temporary basis under Regulation 174 of the Human Medicine Regulations 2012, but this was always intended to be a temporary arrangement. 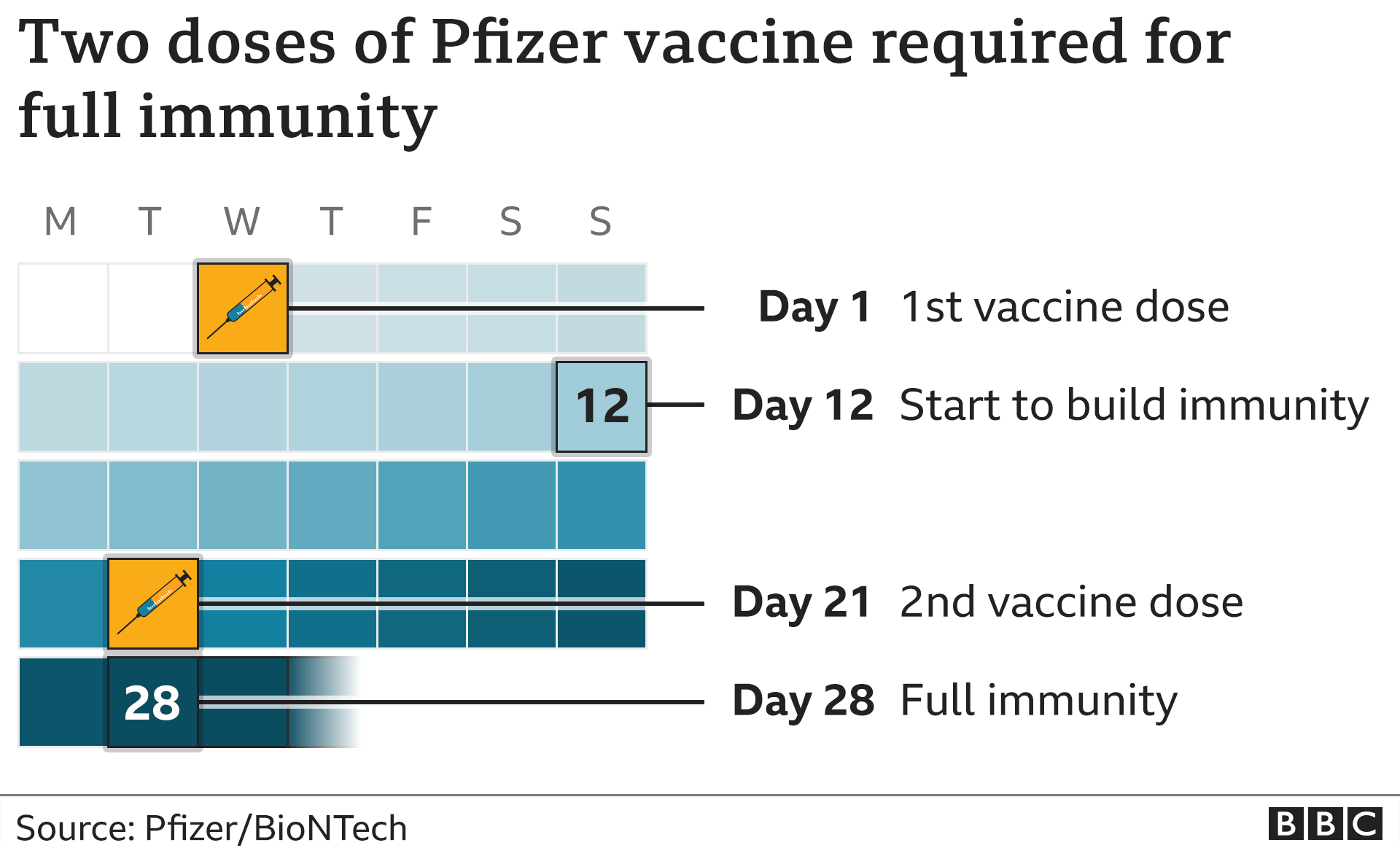
There is currently no GB supply of the fourth presentation and the product information for this will only be published once this supply becomes available.Īll four presentations use the same drug substance but have specific administration requirements and are differentiated by use of different coloured caps on the vials. The product information for the above three presentations can be found on this page.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
